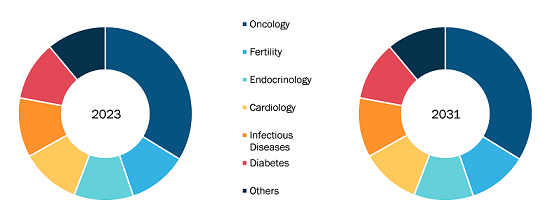
Latin America Chemiluminescence Immunoassay Market
Chemiluminescence immunoassays (CLIA) can detect substances that cannot be identified in radioimmunoassays (RIA) and enzyme-linked immunosorbent assays (ELISA), which is crucial for the early diagnosis of diseases. Chemiluminescence is more rapid, stable, sensitive, specific, and reproducible than ELISA. This detection technique is used in Western blotting, DNA hybridization, forensic science, and CLIA. CLIA is used to detect autoantibodies in biomedical applications such as hemostasis testing, cancer and hepatitis diagnosis, and clinical studies. With the help of CLIA technology, healthcare institutions/facilities can detect and monitor cancer, endocrinological conditions, fertility biomarkers, autoimmune disorders, and infectious diseases, among others. During the COVID-19 pandemic, CLIA was in use to detect anti-SARS-CoV-2 IgG and IgM in patient samples.
The accurate diagnosis of HIV infection requires tests that are performed in a specific sequence or algorithm. CLIA, immunofluorescent assays, and line immunoassays are available for selective HIV antibody detection. Commercial assays based on P24 antigen detection are also available for HIV diagnosis. The US Food and Drug Administration (FDA) has licensed Gen-Probe’s APTIMA HIV-1 qualitative RNA assay to diagnose acute HIV-1 infection using plasma samples. The VITROS HIV Combo is a fourth-generation assay that detects HIV-1 and HIV-2 antibodies as well as the p24 antigen in time less than third-generation assays. The HIV Ag/Ab combo assay is a chemiluminescent microparticle immunoassay (CMIA) used for the simultaneous qualitative detection of the HIV p24 antigen as well as anti-HIV type 1 (HIV-1 group M and group O) and anti-HIV type 2 (HIV-2) antibodies from human serum and plasma (treated with EDTA and heparin) samples. Further, the ARCHITECT HIV Ag/Ab Combo Reagent Kit (2P36) is available for all ARCHITECT i Systems in the market. This combo uses anti-HIV-1 p24 antibodies as reagents to detect the corresponding antigen, thereby enabling the improved and early detection of HIV infection. It can also diagnose HIV-1/HIV-2 conditions in pregnant women and children up to 2 years of age.

In April 2024, Mindray introduced the CL-2600i CLIA analyzer for mid-sized laboratories. The product is capable of processing up to 240 tests per hour, with a large consumable capacity. It incorporates the latest technologies, all within just one square meter of a product, thereby saving space without compromising performance. In November 2023, Medical & Biological Laboratories Co., Ltd., a JSR Life Sciences Company, introduced an automated mono-test chemiluminescence immunoassay analyzer—iStar 500—for the global market. The product is a highly automated mono-test analyzer based on CLIA technology. The compact and integrated design, combined with highly sensitive CLIA technology, makes it an ideal product for emergency diagnosis or small- to medium-sized labs looking for accurate test results. The product can be employed to test parameters associated with conditions such as cardiovascular diseases, reproductive health, liver health, and autoimmunity; moreover, it can detect tumor markers.
Growing Use of Point-of-Care Diagnostics Drives Latin America Chemiluminescence Immunoassay Market Growth
Point-of-care diagnostics (POCD) play a vital role in the healthcare sector. It has gained significant importance for rapid diagnosis of various infectious and life-threatening diseases, such as cancer and diabetes, along with monitoring health conditions. POC devices are efficient diagnostic options to prevent delays in treatment, a crucial aspect in preventing high rates of mortality and transmission of infectious agents. Furthermore, the COVID-19 pandemic created a paradigm shift in the POC testing scenario in Latin America. It has driven research focused on developing rapid and economical testing strategies that can be implemented as point-of-care testing. The COVID-19 pandemic has accelerated the decentralization in point-of-care testing, increasing patient reach outside laboratories, hospitals, and homes enables a faster response to clinical needs.
Scientists and entrepreneurs have made technological advancements to develop the next-generation POC diagnostic devices based on the latest technologies and focus on automation for different applications. Also, companies are developing affordable POC diagnostic products with high specificity and sensitivity. For instance, Fapon, a leading life sciences company, launched its latest chemiluminescence immunoassay system, Shine i8000/9000, in global market at the 2023 AACC exhibition in July 2023. The fully automated high-speed analyzer has an ultra-high throughput of 900 tests per hour that enables its partners to develop premium clinical diagnostic solutions.
Latin America Chemiluminescence Immunoassay Market: Competitive Landscape and Key Developments
Sysmex Corp, Shenzhen Mindray Bio-Medical Electronics Co Ltd, Bio-Rad Laboratories Inc, Merck KGaA, Abbott Laboratories, Thermo Fisher Scientific Inc., F. Hoffmann-La Roche Ltd, DiaSorin SpA, Siemens Healthineers AG, and Getein Biotech Inc. are a few key companies operating in the market. These companies focus on product innovation strategies to meet evolving customer demands, along with maintaining their brand name in the Latin America chemiluminescence immunoassay market.