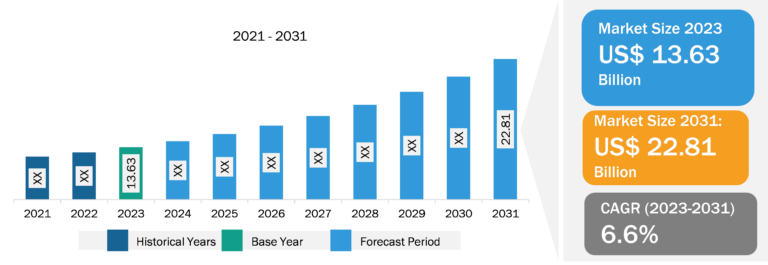
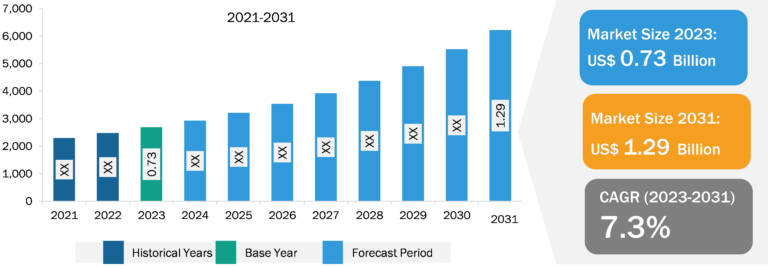
Europe Intra-Articular Injection for Osteoarticular Infiltrations Market
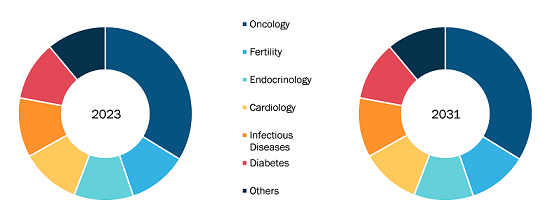
As more people seek to manage their joint pain and improve their quality of life, intra-articular injections have emerged as a highly effective solution due to their targeted approach and relatively quick onset of relief compared to systemic medications. Furthermore, advancements in injection techniques and the development of new therapeutics, such as hyaluronic acid and biologicals, are expanding the range of options available to patients, enhancing efficacy and safety profiles. Recent advancements in hyaluronic acid injection therapy have significantly expanded growth trajectory. Focus has shifted toward hyaluronic acid-based hydrogel therapeutics, which serve as innovative delivery systems for osteoarthritis treatment. Furthermore, the increasing awareness among healthcare professionals and patients regarding the benefits of these injections, combined with increasing investment in healthcare infrastructure and innovation, is facilitating increased access to these treatments. Finally, the emphasis on personalized medicine is leading to tailored treatment plans, making intra-articular injections an attractive option in a comprehensive osteoarthritis management plan. Furthermore, the rising prevalence of osteoarthritis and advancements in injection techniques are noteworthy factors contributing to the expansion of the Europe intra-articular injection for osteoarticular infiltrations market size. On the other hand, the side effects associated with intra-articular injections hinder market growth. Moreover, increasing number of clinical trials is expected to enhance the development of innovative products, which is anticipated to bring new Europe intra-articular injection for osteoarticular infiltrations market trends in the coming years.
Advancements in Injection Techniques Drives Europe intra-articular injection for osteoarticular infiltrations Market Growth
Initially, landmark-guided techniques that had an average accuracy rate of 79% were used for injections, and most health professionals relied on these techniques. With the development of imaging technologies such as ultrasound, CT, and MRI, the detection precision increased drastically. These techniques increased the accuracy rate of intra-articular injection up to 98%. The portable options of ultrasound, CT, and MRI are further expected to enhance the accessibility of such advanced technologies. Thus, with the advancements in imaging techniques, intra-articular injections have become increasingly common in clinical practice. These advanced technologies not only improve the accuracy of intra-articular injections but also help patients in pain management and functional improvement over those receiving landmark-guided injections. Furthermore, image-guided injections are likely to be more cost-effective than landmark-guided injections, possibly due to the enhanced precision of image guidance, resulting in reduced follow-up costs. Therefore, the advancements in image-guided techniques to achieve improved clinical outcomes are contributing to the Europe intra-articular injection for osteoarticular infiltrations market growth.

Europe Intra-Articular Injection for Osteoarticular Infiltrations Market: Competitive Landscape and Key Developments
Bioventus Inc., SEIKAGAKU CORPORATION, Maxigen Biotech Inc., İstem Medical Inc., Sanofi SA, Ferring Pharmaceuticals, Fidia Pharma USA Inc., Biotech Healthcare Group, Bioteck S.p.A., and I+MED are a few key companies operating in the market. These companies focus on product innovation strategies to meet evolving customer demands, along with maintaining their brand name in the Europe intra-articular injection for osteoarticular infiltrations market.
- In June 2019, Fidia’s Hyaluronic Acid from fermentation has been granted the Certificate of Suitability (CEP, R0-CEP 2017-220-Rev 00) to the European Pharmacopeia, Sodium Hyaluronate Monograph, by EDQM (European Directorate for the Quality of Medicines).
- In June 2020, Ono Pharmaceutical Co., Ltd and Seikagaku Corporation disclosed the results from a Phase III clinical study of ONO-5704/SI-613 in Japan in patients with knee osteoarthritis (“the Study”) that were presented at the 93rd Annual Meeting of the Japanese Orthopaedic Association, an on-line annual meeting being held from Thursday June 11 to Monday August 31, 2020. ONO-5704/SI-613 is being jointly developed in Japan by the companies for the treatment of osteoarthritis.
- In May 2021, Ono Pharmaceutical Co., Ltd. and Seikagaku Corporation together has launched JOYCLU 30mg Intra-articular Injection (generic name: diclofenac etalhyaluronate sodium; “JOYCLU”), a joint function improvement agent, for which Seikagaku received the manufacturing and marketing approval in Japan in March 2021.
- In November 2022, Maxigen Biotech Inc. (MBI) signed an exclusive distribution agreement with Mars Medicine Sağlık Ürünleri Limited Şirketi, a large distributor of pharmaceutical products in Turkey, as Maxigen Biotech’s long-term, strategic partner in opening the Turkish market. This partnership aims to provide the best intra-articular injection technology to medical institutions in Turkey by supplying MBI’s products, which offers patients a full range of knee treatment solutions.
- In February 2023, Fidia Pharmaceutical, has entered the Polish pharma market by acquiring the Polish Business of U.S. based Unipharm Polish affiliate. This is another step in strengthening the company’s position on the European market.