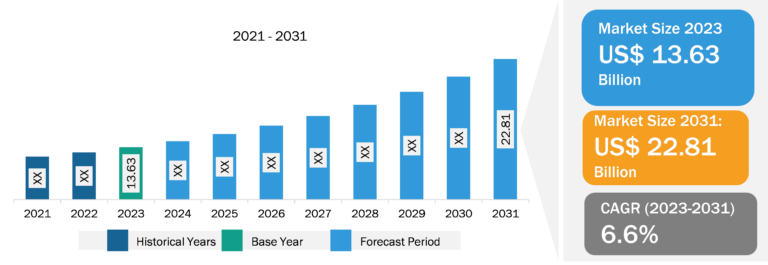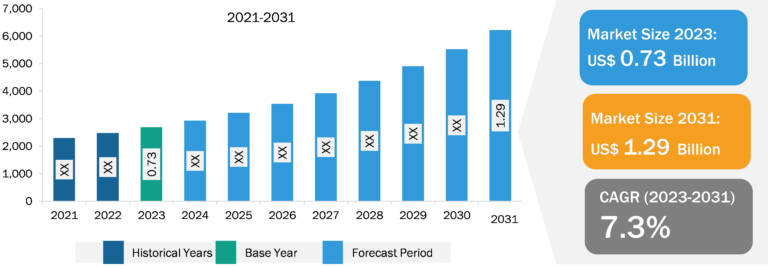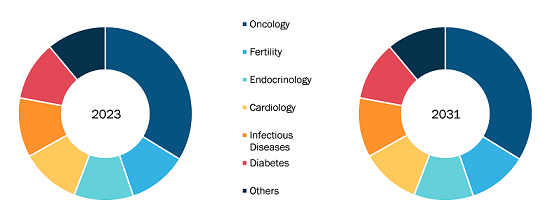
Monocyte Activation Tests Market
Key factors driving the global monocyte activation tests market growth are rise in safety concerns among patients and a surge in demand for safer pyrogen testing methods in end user industries. Technological developments in monocyte activation test methods are expected to propel the overall market growth during the forecast period.
Market Driver
Growing Demand from Medical Device Industry
Monocyte activation tests (MATs) are human cell-based tests to detect and quantify pyrogens such as bacteria, fungi, and viruses. MATs use an ELISA assay to measure cytokine release from treated blood cells. MATs are widely available but rarely used in place of animal-based pyrogen tests for biocompatibility assessment of medical devices. The National Toxicology Program Interagency Center for the Evaluation of Alternative Toxicological Methods (NICEATM) and the PETA International Science Consortium Ltd. (PISC) convened a September 2018 workshop at the National Institutes of Health to elaborate the necessary steps toward implementation of MAT use in medical device testing. According to Luxoft, a DXC Technology Company, medical devices are aiding the digital transformation of healthcare by providing accurate diagnoses, effective treatments, and personalized care through predictive algorithms and patient data analysis. Technological advancements in personalized medicine, implantable devices, smart medical devices, and noninvasive surgery are revolutionizing the overall healthcare industry by offering better care, improved patient outcomes, and reduced costs. Thus, the growth in the medical device industry has increased the demand for monocyte activation test methods, which drives the market. According to our recent analysis, the medical device industry is poised for steady growth, with global annual sales forecast to rise by over 5 percent a year and reach nearly US$800 billion by 2030 . These projections reflect increasing demand for innovative new devices (such as wearables) and services (such as health data), as lifestyle diseases become more prevalent, and economic development unlocks the huge potential in emerging countries – particularly China and India.
Market Trend
Rising Demand for Monocyte Activation Test Methods in Pharmaceutical and Biotechnology Industries
According to the National Library of Medicine, pharmaceuticals are a group of emergent organic compounds that have contributed to the improving quality of life of patients. The pharmaceutical sector is involved in the production, development, and marketing of branded and generic pharmaceuticals. For the first time in 2014, total pharmaceutical revenues across the globe exceeded US$ 1 trillion. The pharmaceutical market has been expanding at an annual rate of 5.8% since 2017. In the same year, worldwide pharmaceutical market revenue was US$ 1,143 billion and reached US$ 1,462 billion in 2021. The monocyte activation test (MAT) is designed to test parenteral drugs, biologics, and medical devices for all classifications of pyrogens. Parenteral-administered pharmaceutical products must be free of pyrogenic (fever-inducing) contamination as these substances might induce a life-threatening systemic response of the patient’s innate immune system. It is to ensure biological products are free of contaminating pyrogenic material prior to administration to patients. Initially, the RPT and the Bacterial endotoxin Test (BET)/Limulus Amebocyte Lysate Assay (LAL) were used as an ex-vivo option. However, stringent regulations adopted for animal testing methods have forced the market participants to develop an alternative method that minimizes the use of such animal test methods. Considering the limitations of the RPT and the BET and increased manufacturing of complex products, the European Pharmacopoeia introduced MAT activation testing methods that simulate the humane immune response and combine the advantages of the RPT (assessment of pyrogenicity beyond gram-negative endotoxin) with the benefits of an invitro method. In contrast to the RPT, MAT can be applied as a fully quantitative test without the use of animals, making it more appropriate for vaccines that are inherently pyrogenic and are physiologically relevant since they use human cells. The MAT testing assays are able to detect blood-derived products, cell-derived products, and biologics and vaccines. The MAT testing methods can also detect a wide testing range of drug products and medical devices as well as products that are unable to undergo in-vivo testing (for example, products containing hyaluronic acid). Such factors have aided the overall monocyte activation tests market in recent years and are expected to follow a similar trend during the forecast period.
Monocyte Activation Tests Market: Segmental Overview
The global monocyte activation tests market is categorized on the basis of source, application, products, and geography. Based on source, the monocyte activation tests market is bifurcated into PMBC and cell line. The PMBC segment held a larger share in 2022 and is expected to continue a similar trend during the forecast period. Based on products, the monocyte activation tests market is bifurcated into MAT kits and reagents. The MAT kits segment held a larger share in 2022 and is expected to continue a similar trend during the forecast period. On the other hand, the reagents segment is anticipated to record a higher CAGR over the forecasted period. Based on application, the monocyte activation tests market is segmented into drug development, vaccine development, medical devices, and others (research, etc.). The drug development segment captured the largest share in 2022 and is expected to witness the same trend during the forecast period.
Monocyte Activation Tests Market: Competitive Landscape and Key Developments
Merck KGaA, Darmstadt, Germany and/or its affiliates; Charles River Laboratories International, Inc.; Thermo Fisher Scientific; Sanquin; and Lonza Group are among the leading companies operating in the monocyte activation tests market.
In October 2023, Lonza launched two new rapid monocyte activation test (MAT) systems, the PyroCell MAT Rapid System and PyroCell MAT Human Serum (HS) Rapid System, to streamline and ease rabbit-free pyrogen testing. The systems will replace Lonza’s traditional MAT system kit offerings, and the newly launched products contain the new PeliKine Human IL-6 Rapid ELISA Kit that minimizes hands-on time and reduces time-to-results from two days to two hours. The new tests give pharmaceutical and biotechnology manufacturers easier MAT testing options for product safety testing while helping to reduce the reliance on animals.