Anesthesia Monitoring Devices Market
Product innovation plays a crucial role in shaping the market landscape of anesthesia monitoring devices, offering substantial growth, advancement, and enhanced patient care. The FDA officially authorized the PMD-200 monitor for sale in the US in March 2023. The device, developed by Medasense Biometrics, measures a patient’s nociception level (NOL), or physiological reaction to pain, by tracking their heart rate, blood pressure, perspiration, and movement via a wearable finger probe. The monitor then uses machine learning to analyze the data and calculate the patient’s pain threshold. Patients who are totally unconscious during surgery are essentially voiceless, as their NOL level speaks for them. The goal is to provide personalized care for every patient. The continuous evolution of technology, coupled with the increasing emphasis on patient safety, precision anesthesia delivery, and perioperative monitoring, drives innovation in anesthesia monitoring devices toward more advanced, efficient, and user-friendly solutions. Innovations in anesthesia monitoring encompass a wide range of developments, including portable monitoring systems, wireless connectivity for seamless data transmission, integration with electronic health records (EHRs) for streamlined documentation, advanced algorithms for data analysis and interpretation, and customizable monitoring parameters tailored to specific patient needs and surgical contexts. These innovations improve the accuracy and reliability of vital sign monitoring and enhance decision-making, anesthesia management strategies, and clinical outcomes for patients undergoing surgical procedures. Furthermore, integrating innovative features, such as artificial intelligence (AI)-driven analytics, cloud-based monitoring platforms, and smart alarms for early detection of anomalies, enables a proactive approach to anesthesia monitoring, risk mitigation, and quality improvement initiatives. Therefore, increasing product innovation is boosting the anesthesia monitoring devices market size.
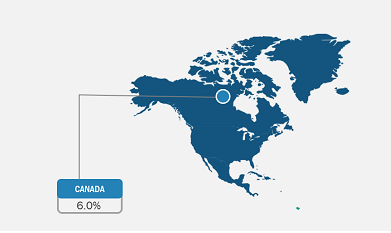
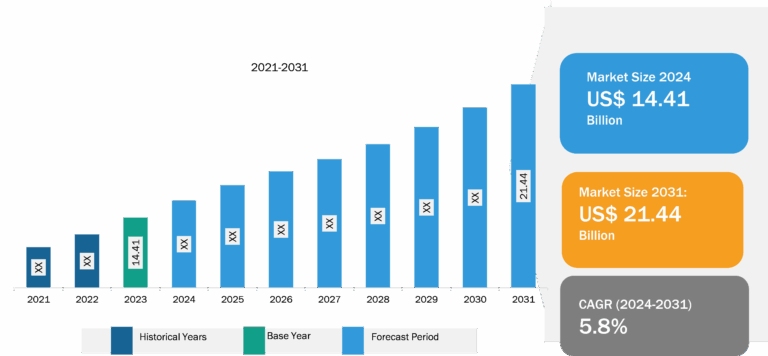
The anesthesia monitoring devices market analysis has been carried out by considering the following segments: product, advanced anesthesia monitors, technology, and end user. By product, the market is classified into basic anesthesia monitors, integrated anesthesia workstations, and advanced anesthesia monitors. Based on advanced anesthesia monitors, the market is segmented into depth of anesthesia, anesthesia gas, and standalone capnograph monitors. In terms of technology, the market is classified into bispectral index, auditory evoked potential, patient state index, cortical activity state index, and others. Based on end user, the market is classified into hospitals, clinics, and ambulatory surgical centers. The scope of the anesthesia monitoring devices market report includes North America (the US, Canada, and Mexico), Europe (the UK, Germany, France, Italy, Spain, and the Rest of Europe), Asia Pacific (China, Japan, India, Australia, South Korea, and the Rest of Asia Pacific), the Middle East & Africa (the UAE, Saudi Arabia, South Africa, and the Rest of Middle East & Africa), and South & Central America (Brazil, Argentina, and the Rest of South & Central America). In 2022, North America held the largest anesthesia monitoring devices market share. In North America, the US holds the largest market share in the world. Market growth in this country is ascribed to the increase in the prevalence of inflammatory diseases, the presence of major market players, continued advancements in the healthcare sector, and access to innovative anesthesia digital monitoring device systems. The market for anesthetic monitoring devices has also grown due to an increase in procedures performed to treat chronic illnesses. For example, according to the data published by Cedars-Sinai in January 2022, over 300,000 coronary artery bypass graft surgeries, often known as bypass surgeries, were performed successfully in the US.
Furthermore, the anesthesia monitoring devices market analysis is carried out by identifying and evaluating key players in the market across different regions.
Anesthesia Monitoring Devices Market: Competitive Landscape and Key Developments
Mindray Medical International Limited; Drägerwerk AG; GE Healthcare; Koninklijke Philips N.V.; Schiller AG; Criticare Systems, Inc.; HEYER Medical AG; Infinium Medical, Inc.; Siare Engineering International Group S.r.l.; and Nihon Kohden Corporation are a few key companies operating in the anesthesia monitoring devices market. Market players adopt product innovation strategies to meet evolving customer demands, maintaining their brand image in the anesthesia monitoring devices market.